Quality-adjusted time without symptoms of disease or toxicity and quality-adjusted progression-free survival with niraparib maintenance in first-line ovarian cancer in the PRIMA trial

Use of First-Line PARP Inhibitors in Ovarian Cancer | Published in healthbook TIMES Oncology Hematology
Niraparib in Patients With Newly Diagnosed Advanced Ovarian BRCAm Cancer: A Post Hoc Analysis of the PRIMA/ENGOT-OV26/GOG-3012 T

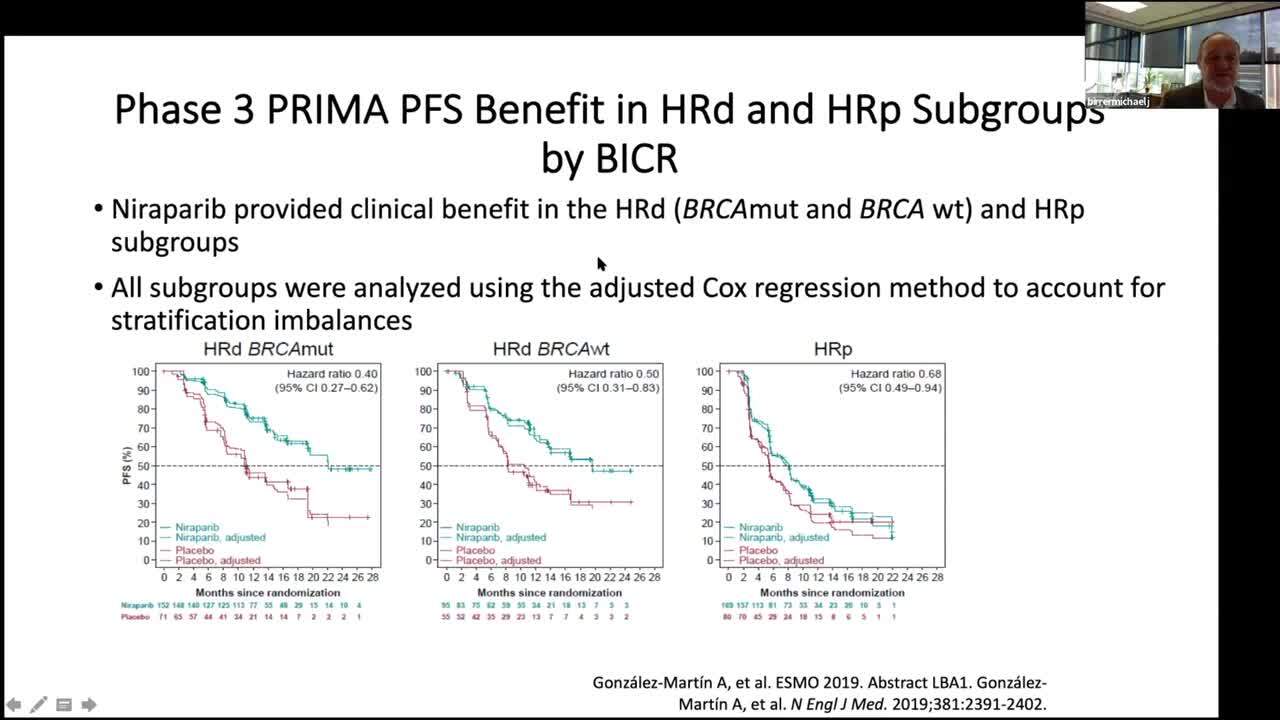
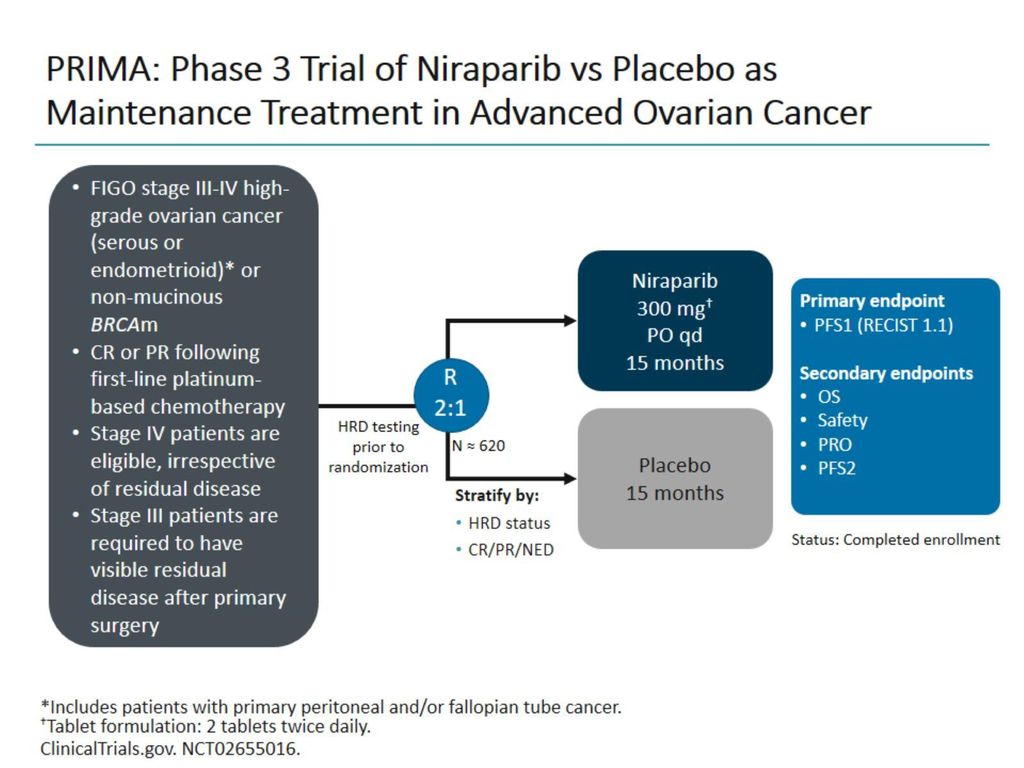
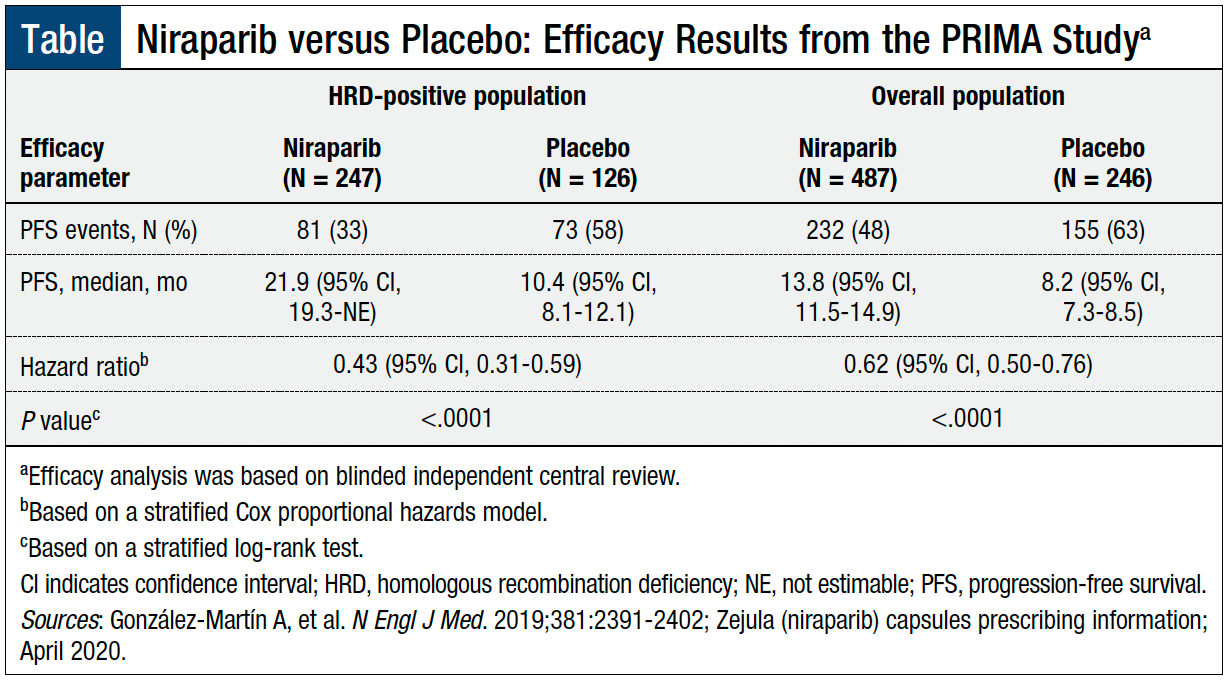
Stephen V Liu, MD on X: "#ESMO19 Practice changing study presented by Antonio Gonzalez Martin. PRIMA study of niraparib in patients with ovarian cancer responding to 1L platinum based chemo. Primary endpoint
Zejula (Niraparib) First PARP Inhibitor Approved for First-Line Maintenance Therapy in All Women with Advanced Ovarian Cancer, Regardless of Biomarker Status

Efficacy and safety of niraparib as maintenance treatment in patients with newly diagnosed advanced ovarian cancer using an individualized starting dose (PRIME Study): A randomized, double-blind, placebo-controlled, phase 3 trial (LBA 5) -

Progression-free survival and safety at 3.5 years of follow-up: results from the randomised phase 3 PRIMA/ENGOT-OV26/GOG-3012 trial of niraparib maintenance treatment in patients with newly diagnosed ovarian cancer - European Journal of